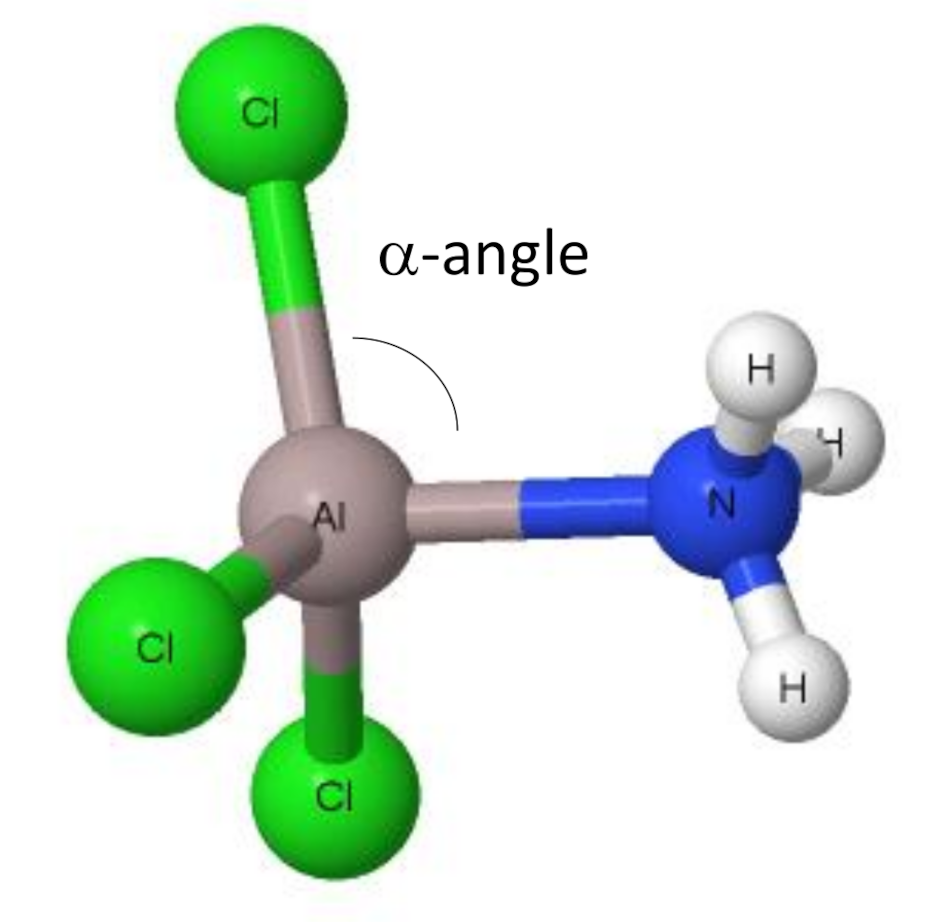
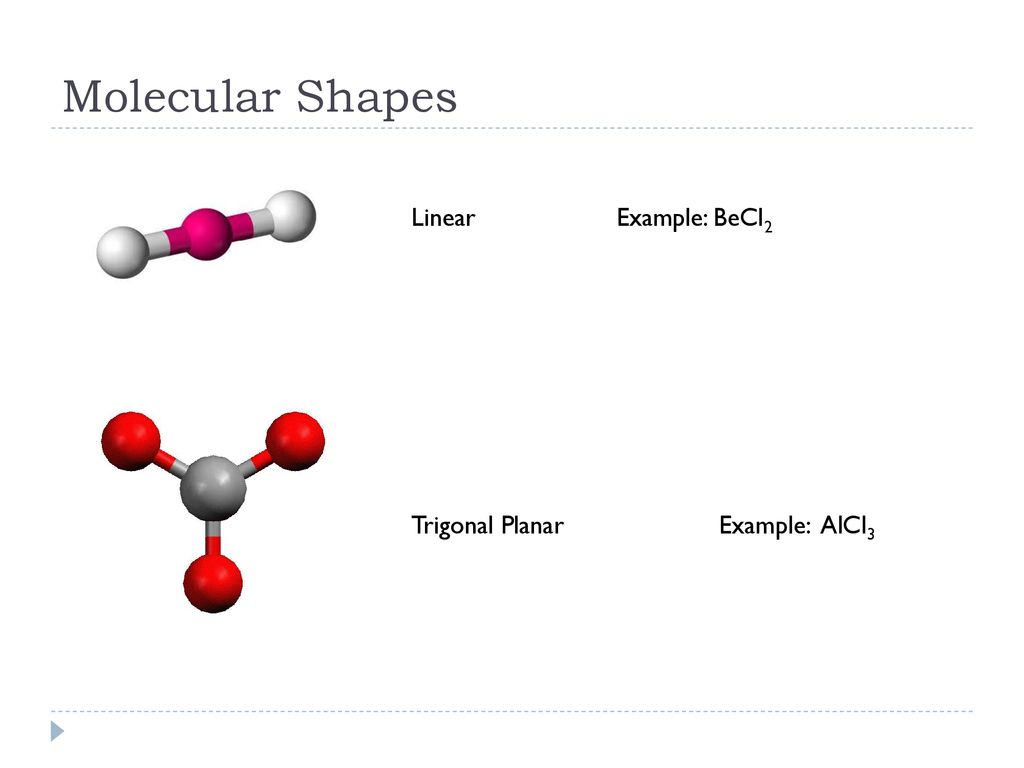
Aluminum chloride (AlCl3) is a fascinating compound with unique molecular geometry that plays a crucial role in various chemical processes and industries. Its structure and properties make it an essential subject of study for chemists and researchers worldwide. Understanding the molecular geometry of AlCl3 is vital for anyone interested in inorganic chemistry or chemical engineering.
The molecular geometry of AlCl3 is not only important for theoretical understanding but also for its practical applications. From catalysis to material science, AlCl3's geometry determines its reactivity and functionality in different environments. This article will delve into the intricate details of AlCl3's molecular geometry, offering insights into its structure, bonding, and applications.
Whether you're a student, researcher, or professional in the field of chemistry, this guide aims to provide a comprehensive overview of AlCl3 molecular geometry. By the end of this article, you'll have a solid understanding of how AlCl3's structure influences its properties and applications, equipping you with valuable knowledge for further exploration.
Table of Contents:
- Introduction to AlCl3 Molecular Geometry
- Understanding the Structure of AlCl3
- Bonding in AlCl3
- The Shape of AlCl3
- Properties of AlCl3
- Applications of AlCl3
- Variations in AlCl3 Molecular Geometry
- Comparison with Other Compounds
- Experimental Determination of AlCl3 Geometry
- Future Research Directions
Introduction to AlCl3 Molecular Geometry
Why Study AlCl3 Molecular Geometry?
Aluminum chloride (AlCl3) is a well-known inorganic compound with a variety of industrial applications. Its molecular geometry is critical to understanding its behavior in chemical reactions and its role as a catalyst. By studying the molecular geometry of AlCl3, chemists can predict how it will interact with other substances and optimize its use in various processes.
Historical Context
The study of AlCl3 molecular geometry dates back to the early 20th century when chemists began exploring the properties of inorganic compounds. Over the years, advancements in spectroscopy and computational chemistry have provided deeper insights into its structure and behavior. Today, AlCl3 remains a key compound in both academic research and industrial applications.
Understanding the Structure of AlCl3
Aluminum chloride exists in two primary forms: monomeric (AlCl3) and dimeric (Al2Cl6). The structure of AlCl3 depends on its phase and environment. In the gas phase, AlCl3 adopts a trigonal planar geometry, while in the solid state, it forms a dimeric structure due to strong intermolecular forces.
Monomeric vs. Dimeric Structure
- Monomeric Structure: In the gas phase, AlCl3 exists as a single molecule with a trigonal planar geometry.
- Dimeric Structure: In the solid state, AlCl3 molecules combine to form Al2Cl6 dimers, which exhibit a tetrahedral geometry around each aluminum atom.
Bonding in AlCl3
The bonding in AlCl3 involves covalent bonds between aluminum and chlorine atoms. Aluminum, being a metal, shares its three valence electrons with three chlorine atoms, forming a stable compound. The nature of these bonds and their influence on molecular geometry is a key aspect of understanding AlCl3's properties.
Electron Deficiency
Aluminum is electron-deficient in AlCl3, meaning it has an incomplete octet. This deficiency leads to the formation of dimers in the solid state, where aluminum atoms share electrons to achieve a more stable configuration. This phenomenon is crucial in determining the overall geometry of AlCl3.
The Shape of AlCl3
The molecular geometry of AlCl3 is primarily determined by the arrangement of its constituent atoms. In the gas phase, AlCl3 exhibits a trigonal planar shape, with bond angles of approximately 120 degrees. In the solid state, the dimeric structure results in a more complex geometry, with tetrahedral arrangements around each aluminum atom.
Factors Influencing Shape
- Electron Pair Repulsion: The VSEPR theory explains the trigonal planar geometry of AlCl3 in the gas phase.
- Intermolecular Forces: In the solid state, strong intermolecular forces lead to the formation of dimers with tetrahedral geometry.
Properties of AlCl3
AlCl3's molecular geometry directly influences its physical and chemical properties. Some key properties include:
- Boiling Point: AlCl3 has a relatively low boiling point due to its molecular structure.
- Reactivity: The trigonal planar geometry of AlCl3 makes it highly reactive, particularly as a Lewis acid.
- Solubility: AlCl3 is soluble in polar solvents, which is influenced by its molecular geometry and bonding characteristics.
Applications in Catalysis
AlCl3's unique geometry and reactivity make it an excellent catalyst in various chemical reactions, particularly in Friedel-Crafts alkylation and acylation processes.
Applications of AlCl3
Industrial Uses
AlCl3 finds extensive use in industries such as:
- Petrochemicals: As a catalyst in the production of aromatic compounds.
- Pharmaceuticals: In the synthesis of drugs and other organic compounds.
- Material Science: For the production of aluminum-based materials and composites.
Research Applications
In academic research, AlCl3 is used to study molecular interactions, catalytic mechanisms, and material properties. Its well-defined geometry makes it an ideal model compound for theoretical studies.
Variations in AlCl3 Molecular Geometry
While the basic geometry of AlCl3 is well-established, variations can occur under different conditions. For example, changes in temperature, pressure, or solvent environment can influence its structure and behavior.
Influence of External Factors
- Temperature: Higher temperatures can favor the monomeric form of AlCl3.
- Pressure: Increased pressure may promote dimer formation.
- Solvent: Polar solvents can stabilize the monomeric form, while nonpolar solvents favor dimers.
Comparison with Other Compounds
AlCl3's molecular geometry can be compared to other aluminum halides, such as AlF3 and AlBr3. While these compounds share similar structures, their properties differ due to variations in bond strength and electronegativity.
Key Differences
- AlF3: Exhibits a more stable octahedral geometry due to the smaller size and higher electronegativity of fluorine.
- AlBr3: Similar to AlCl3, but with slightly weaker bonds due to the larger size of bromine atoms.
Experimental Determination of AlCl3 Geometry
Modern techniques such as X-ray diffraction, nuclear magnetic resonance (NMR), and computational modeling are used to determine the molecular geometry of AlCl3. These methods provide precise information about bond lengths, angles, and overall structure.
Computational Methods
Molecular modeling software, such as Gaussian and VASP, allows researchers to simulate the geometry of AlCl3 under various conditions. These simulations are invaluable for predicting behavior and optimizing applications.
Future Research Directions
Despite extensive research, there is still much to learn about AlCl3 molecular geometry. Future studies may focus on:
- Exploring new applications in nanotechnology and material science.
- Investigating the effects of novel catalysts on AlCl3 reactions.
- Developing more efficient methods for determining molecular geometry under extreme conditions.
Conclusion
In conclusion, the molecular geometry of AlCl3 is a critical factor in determining its properties and applications. From its trigonal planar structure in the gas phase to its dimeric form in the solid state, AlCl3's geometry plays a vital role in its reactivity and functionality. By understanding the nuances of AlCl3's molecular geometry, chemists can unlock new possibilities in catalysis, material science, and beyond.
We invite you to share your thoughts and questions in the comments section below. Additionally, explore other articles on our site to deepen your knowledge of chemistry and related fields. Together, we can continue to advance our understanding of the fascinating world of molecular geometry.